Plasticity and adaptation in the visual cortex
Neurons in the mammalian visual cortex are tuned to respond to visual stimuli such as contour orientation, motion, direction, and speed. Preference for orientation in orientation columns is considered relatively stable in the primary visual cortex (V1) as an emergent property that is established early in the life, following the so-called critical period.
Studies from various laboratories have shown that in a fully mature brain, neuronal network restructures itself beyond the postnatal critical period that follows birth. Recent investigations revealed the ability of visual neurons to respond to different stimuli conditions (deprivation or imposition) by changing their optimal properties acquired after birth. This adaptation of neurons for visual perception suggests the existence of neuronal plasticity in adults, hence a mature brain.
Adaptation-induced-plasticity of orientation in primary visual cortex is characterized as the ability of cortical neurons to change their preferred orientation following a long or short exposure to a non-preferred orientation for the primary visual cortex neurons in cats, e.g. Long adaptation leads to the shift of orientation tuning towards attractive direction. In a similar fashion, repetitive adaptation to a non-preferred spatial frequency reveals the spatial frequency tuning shifts in cat visual cortex. In general, imposing a particular stimulus induces instructive process to modify neuronal properties, for example, when in the visual cortex of awake mice a single orientation grating stimulus is repeatedly presented; it leads to augmentation of responses evoked exclusively by testing stimulus, that is, the experience led to enhancement of response. In experiments where animals are anesthetized the shifts of peaks of tuning curves following adaptation are not attributed to attention modulations. Consequently, these shifts result from basic neuronal processes outside the frame of attentional processes that might impact response magnitudes.
Adaptation studies in recent years have presented a more complex picture where prolonged exposure to a non-preferred orientation has shown modifications in neurons’ preferred orientations. After adaptation to a non preferred orientation, obtained tuning curve for the new preferred orientation (after adaptation) can shift in two directions relative to the original preferred orientation: attractive or repulsive. An attractive shift is a shift of the tuning curve towards the adapting orientation. On the other hand; a repulsive shift is a shift of the tuning curve in the opposite side of the adapting orientation.
Attractive shifts are more frequent than repulsive shifts in longer adaptation durations (≥ 6 min). Repeated or prolonged exposure to an adapter diminished neuronal responses evoked by the original optimal properties, furthermore in parallel, if it is the neuron’s preferred stimulus. Optical imaging investigations in recent years have also revealed the impact of adaptation-induced-plasticity, showing that orientation maps in V1 can be modified by imposing one particular orientation.
Studies from various laboratories have shown that in a fully mature brain, neuronal network restructures itself beyond the postnatal critical period that follows birth. Recent investigations revealed the ability of visual neurons to respond to different stimuli conditions (deprivation or imposition) by changing their optimal properties acquired after birth. This adaptation of neurons for visual perception suggests the existence of neuronal plasticity in adults, hence a mature brain.
Adaptation-induced-plasticity of orientation in primary visual cortex is characterized as the ability of cortical neurons to change their preferred orientation following a long or short exposure to a non-preferred orientation for the primary visual cortex neurons in cats, e.g. Long adaptation leads to the shift of orientation tuning towards attractive direction. In a similar fashion, repetitive adaptation to a non-preferred spatial frequency reveals the spatial frequency tuning shifts in cat visual cortex. In general, imposing a particular stimulus induces instructive process to modify neuronal properties, for example, when in the visual cortex of awake mice a single orientation grating stimulus is repeatedly presented; it leads to augmentation of responses evoked exclusively by testing stimulus, that is, the experience led to enhancement of response. In experiments where animals are anesthetized the shifts of peaks of tuning curves following adaptation are not attributed to attention modulations. Consequently, these shifts result from basic neuronal processes outside the frame of attentional processes that might impact response magnitudes.
Adaptation studies in recent years have presented a more complex picture where prolonged exposure to a non-preferred orientation has shown modifications in neurons’ preferred orientations. After adaptation to a non preferred orientation, obtained tuning curve for the new preferred orientation (after adaptation) can shift in two directions relative to the original preferred orientation: attractive or repulsive. An attractive shift is a shift of the tuning curve towards the adapting orientation. On the other hand; a repulsive shift is a shift of the tuning curve in the opposite side of the adapting orientation.
Attractive shifts are more frequent than repulsive shifts in longer adaptation durations (≥ 6 min). Repeated or prolonged exposure to an adapter diminished neuronal responses evoked by the original optimal properties, furthermore in parallel, if it is the neuron’s preferred stimulus. Optical imaging investigations in recent years have also revealed the impact of adaptation-induced-plasticity, showing that orientation maps in V1 can be modified by imposing one particular orientation.
Relationships between the representations of orientations in the primary and secondary areas of the visual cortex of the cat by intrinsic optical imaging method.

Problem:
Orientations are represented in the primary areas (V1) and secondary (V2) columns, the neurons in a column respond to a specific orientation. However, the way in which visual information is integrated from one area to another is partially unknown. To try to understand the relationships between areas V1 and V2, we present a unique non preferred stimulus, i.e. orientation to adult anesthetized cats (this process is called adaptation).
We know already that adaptation process changes the representation of columns in V1 (Bachatene et al. NeuroReport 2012; Ghisovan et al., PLoS One, 2008, Neurosci. 2009). Given the many reciprocal connections between V1 and V2, we want to know how adaptation affects the representation of orientations in V2. Do V2 representations vary similarly to V1? Do they vary in the same proportions?
Methodology:
We present series of eight different orientations to anesthetized cats while we record with a CCD camera images of the cortical area corresponding to V1 and V2.
We record from the beginning of the experiment (control). Then we choose a direction and present for 3 or 12 minutes (adaptation). Immediately after adaptation, we make a second recording. After 2 hours period, we make a third recording (recovery). The images of these recordings are then denoised and maps guidelines for the three recordings are generated (see figure).
Orientation map control. Each color corresponds to a given direction (in degrees). Pixels are represented by the color of the orientation for which they respond maximally. A pixel covers about 13 microns of cortical surface.
Orientations are represented in the primary areas (V1) and secondary (V2) columns, the neurons in a column respond to a specific orientation. However, the way in which visual information is integrated from one area to another is partially unknown. To try to understand the relationships between areas V1 and V2, we present a unique non preferred stimulus, i.e. orientation to adult anesthetized cats (this process is called adaptation).
We know already that adaptation process changes the representation of columns in V1 (Bachatene et al. NeuroReport 2012; Ghisovan et al., PLoS One, 2008, Neurosci. 2009). Given the many reciprocal connections between V1 and V2, we want to know how adaptation affects the representation of orientations in V2. Do V2 representations vary similarly to V1? Do they vary in the same proportions?
Methodology:
We present series of eight different orientations to anesthetized cats while we record with a CCD camera images of the cortical area corresponding to V1 and V2.
We record from the beginning of the experiment (control). Then we choose a direction and present for 3 or 12 minutes (adaptation). Immediately after adaptation, we make a second recording. After 2 hours period, we make a third recording (recovery). The images of these recordings are then denoised and maps guidelines for the three recordings are generated (see figure).
Orientation map control. Each color corresponds to a given direction (in degrees). Pixels are represented by the color of the orientation for which they respond maximally. A pixel covers about 13 microns of cortical surface.
Neuronal encoding
Brain is a vast jungle of connections and the complex behaviour of animals is related to the complex activity of this organ. To understand the structural and functional dynamics (connectomics) of brain is a central challenge for neuroscientists.Visual cortex is perhaps the most studied area of the brain, and has provided us numerous insights into working of the brain. The theme of this research is “Encoding the Visual stimuli by forming neuronal networks”, as how neurons encode a stimulus has largely been an unresolved problem yet, though with advances in technology it does not seem a distant goal that this corollary (brain) which drives us all would remain a quandary.
The main objectives that we address :
1.How visual neurons link with each other in response to various stimulus features( e.g. orienation, spatial frequency)
2.Simultaion of the in vivo-results. An illustaration of network of simultaneously recorded neurons from cat primary visual cortex in response to different orientation gratings :
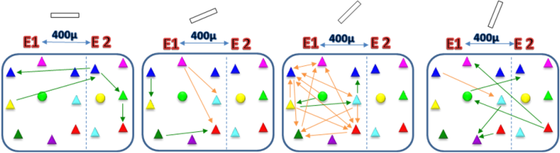
Figure Network between simultaneously recorded neurons using a tungsten multielectrode (400 microns apart). Neuronal network is selective with changing orientation. The functional connections between the same neurons change for different orientations (sine wave drifting gratings - shown at the top of network box). Arrows represent the functional connections as revealed by crosscorrelograms. Orange arrows represent maintained connections and green arrows correspond to new connections. Triangle: Pyramidal cell; Circle: Interneuron. E1: First electrode; E2: Second electrode.
Understanding how the brain works could help the humanity immensely. This project which will reflect on the neuronal communication and codes governing the visual stimuli will be an essential step further to reveal and comprehend the brain dynamics not only for fundamental reasons but which applies to various medical reasons equally e.g. the brain disorders, where often the neuronal wiring goes haywire.
The main objectives that we address :
1.How visual neurons link with each other in response to various stimulus features( e.g. orienation, spatial frequency)
2.Simultaion of the in vivo-results. An illustaration of network of simultaneously recorded neurons from cat primary visual cortex in response to different orientation gratings :
Figure Network between simultaneously recorded neurons using a tungsten multielectrode (400 microns apart). Neuronal network is selective with changing orientation. The functional connections between the same neurons change for different orientations (sine wave drifting gratings - shown at the top of network box). Arrows represent the functional connections as revealed by crosscorrelograms. Orange arrows represent maintained connections and green arrows correspond to new connections. Triangle: Pyramidal cell; Circle: Interneuron. E1: First electrode; E2: Second electrode.
Understanding how the brain works could help the humanity immensely. This project which will reflect on the neuronal communication and codes governing the visual stimuli will be an essential step further to reveal and comprehend the brain dynamics not only for fundamental reasons but which applies to various medical reasons equally e.g. the brain disorders, where often the neuronal wiring goes haywire.

